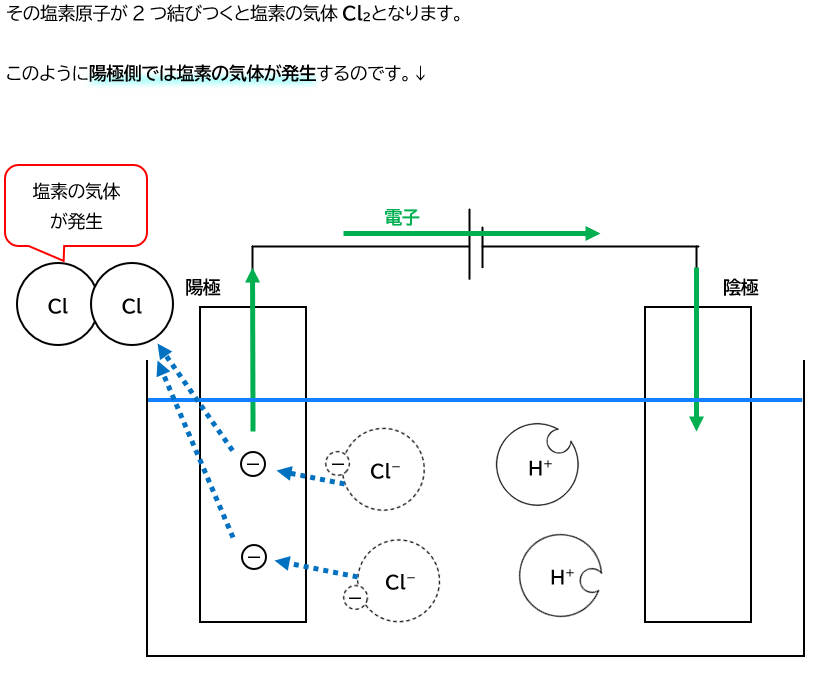
塩酸の電気分解
電気分解とは、電気を通すことによって行われる分解のことです。
電気分解 =「溶液に電気を流すと、溶液中のイオンが電子を得たり離し
たりして原子に戻る反応」というイメージを持っておきましょう。













次回は、塩化銅水溶液の電気分解についてです!
塩酸の電気分解
電気分解とは、電気を通すことによって行われる分解のことです。
電気分解 =「溶液に電気を流すと、溶液中のイオンが電子を得たり離し
たりして原子に戻る反応」というイメージを持っておきましょう。














次回は、塩化銅水溶液の電気分解についてです!